The latest epilepsy news

“Fit and healthy” footballer died of SUDEP, inquest rules
Kami Kountcheva | Inquest finds that 27-year-old footballer Emily Wilcock died of sudden unexpected death in epilepsy (SUDEP) days after stopping her medication.
The latest epilepsy news

Olivia Colman launches one-of-a-kind merch with Epilepsy Action to raise funds
Kami Kountcheva | Olivia Colman launches merchandise with Epilepsy Action to help raise vital funds for epilepsy.
Sponsored news

A New Option for Epilepsy Treatment: UK’s First EASEE® Implant
Precisis | More information about the EASEE® implant for focal epilepsy.
The latest epilepsy news
Link between gabapentin and dementia – study
Kami Kountcheva | New US study finds link between gabapentin use and a higher risk of dementia.
The latest epilepsy news
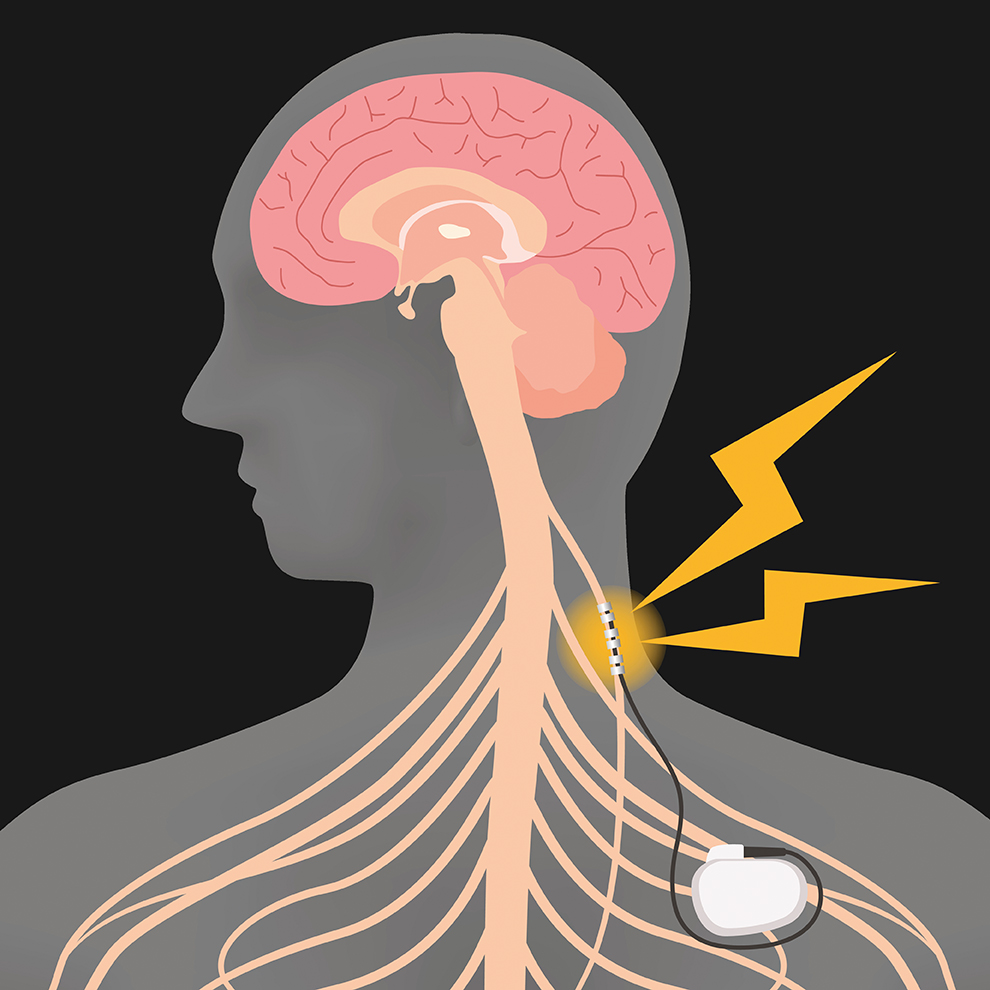
Trial investigating long-term VNS effectiveness launches
Kami Kountcheva | A new, seven-year project from the University of Liverpool will investigate the long-term effectiveness of VNS in refractory epilepsy.
Features

Valproate redress – five years on, still waiting
Kami Kountcheva | Three women, Becci Smart, Susan Jamison and Susan Cole describe what life has been like for their families affected by the valproate…
The latest epilepsy news

PIP U-turn in welfare bill shortly before it is voted in by Parliament
Kami Kountcheva | Parliament has passed the government’s welfare bill after another big U-turn on PIP, meaning no changes will be made to this benefit…
The latest epilepsy news

PIP safe for current claimants after government welfare bill concession
Kami Kountcheva | The government has made a concession on its welfare bill, which means current PIP claimants won’t be affected by the changes to…
The latest epilepsy news

Neuro Survey reveals financial and mental health struggles in epilepsy
Kami Kountcheva | The My Neuro Survey has found a lack of access to mental health services and financial struggles among people with epilepsy.
Page 1 of 64

